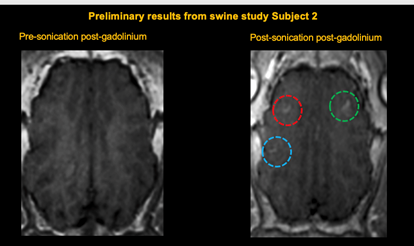
Mountain View, Calif. – Cordance Medical, developer of the NeuroAccess™ device, has demonstrated precise multi-site openings of the blood-brain barrier in animals. The study is utilizing a pig model to assess the safety and efficacy of the NeuroAccess™ device and its ability to open the blood-brain barrier in a controlled and temporary manner. In the studies conducted so far and in several subjects, the NeuroAccess™ treatment resulted in openings of the blood-brain with no evidence of neurologic damage as evidenced by MRI imaging.
“Our most recent preclinical study highlights the first functional studies of our NeuroAccess™ device, which we plan to use in human studies later this year,” said Bhaskar Ramamurthy, Ph.D., co-founder & CTO of Cordance Medical. “Using Cordance-patented beamforming technology, multiple cranial sites across several subjects were observed through gadolinium-contrast magnetic resonance imaging (MRI), without evidence of damage.”
“These results represent a major milestone in the development of our NeuroAccess device,” said Ryan Dittamore, CEO of Cordance Medical. “One of our differentiating features is in the ability to deliver ultrasound therapy non-invasively, to multiple sites in the brain, with safety, precision and accuracy. This successful demonstration supports our development efforts to utilize NeuroAccess™ in patients with brain tumors, Alzheimer’s, and other neurologic diseases with a device that maximizes patient comfort and physician workflow.”
Cordance Medical is currently in the preclinical stage of development for its NeuroAccess™ device, focusing on treating brain tumors. The company is also exploring additional indications for its technology, including the treatment of neurodegenerative disorders and central nervous system infections.
